Emergency managers never plan to resolve a pandemic with the use of a vaccine. This is because a vaccine for a new virus takes three to five years to fully develop, even when pushed forward with emergency urgency, to ensure that the vaccine will be both safe and effective. By the time a safe and effective vaccine is ready to go, the virus causing the pandemic has become endemic and society has moved on with their lives.
The reason it takes a long time to develop a vaccine is because viruses and vaccines are complex. Is a vaccine even possible? What are the side effects? Is the vaccine safe and effective?
A constantly shifting and changing virus may be impossible to resolve with a vaccine. In such a case, no matter how “safe” a vaccine may be, if not “effective” then the vaccine is a waste of time and money and will never resolve a pandemic. The common cold to this day does not have a vaccine for that very reason.
Safe and Effective?
It is important to define the two terms – “safe” and “effective”.
In the past a vaccine was deemed to be “safe” if the risk from taking the vaccine was markedly less than the risk of catching the virus. No vaccine has ever been 100% risk free for everyone.
In the past, for a vaccine to be “effective” we assumed, once vaccinated, a person’s immune system would be able to fight off future infections from the virus such that:
- the person would not feel ill,
- the person would not transmit the virus to others,
- the person would not need hospitalization, and
- the person would not die from the virus.
To achieve the two outcomes, safe and effective, a vaccine needs to be tested across all sectors of society, that will be impacted by the virus, before the vaccine is used in those groups. Testing needs to be comprehensive to look at differences in reactions based on:
- age
- being immuno-compromised
- pre-existing single medical conditions
- pre-existing multiple medical conditions
- race
- genetic disorders
- pregnancy / lactation / reproduction
- prior infection by the virus
- sex / Gender
This testing develops the parameters or factors that ensure full informed consent for the use of the vaccine. This is based on a cost benefit analysis (CBA), or risk versus reward evaluation. In simple terms, is the risk of the vaccine to the individual or group of individuals in question less than the risk of the virus to the same group.
In rare cases, blanket approval for all members of a society may be possible, with a completely uniform cost benefit for all members of the society. That approval should only be given following the focused testing described above and after extensive Vaccine Adverse Event Records System (VAERS) styled findings are published from the tests.
Emergency approval of vaccines should only be given group by group as the testing above is completed. Test results for one group should never be assumed to be true for all groups that were not included in the testing. Further, steps in the testing protocols should not be skipped. There is a reason for them.
What reason? Let’s go back to the terms “safe” and “effective” with respect to the COVID-19 Pandemic.
Safe? To date, the testing on the “vaccines” we are now employing have not undergone the normal or even emergency level of testing required for approval (safe to use) across all sectors of our society. In fact, certain risk sectors have intentionally not been included in the 12-week and 6-month trials. Regardless, the vaccines are being administered to all sectors of our society.
Reported VAERS data has shown that there have been more severe outcomes from these vaccines in one year, than in 40 years of ALL vaccines previously approved. More ominously, the VAERS data shows that the deaths caused by these vaccines is more than double the deaths in ALL vaccines approved in the last 40 years. Yet, despite these horrendous results, the official position continues to be that vaccines are the only way out of this pandemic.
Effective? These “vaccines” are not stopping people from catching the virus and feeling ill; vaccinated people are still transmitting the virus to others; vaccinated people may still need hospitalization and may die from the virus. The worldwide results indicate vaccines are not “effective” according to previous definitions.
Below is the data for one Province in Canada (Alberta), dated January 2022. They show that over 78% of active cases are double or triple vaccinated, and that 59% of hospitalized COVID-19 patients are double or triple vaccinated. In Israel and the UK, which have had high vaccination rates previous to Alberta, the case and hospitalization numbers are even greater. Yet again, the official policy continues to be that these vaccines are the only way out of this pandemic.
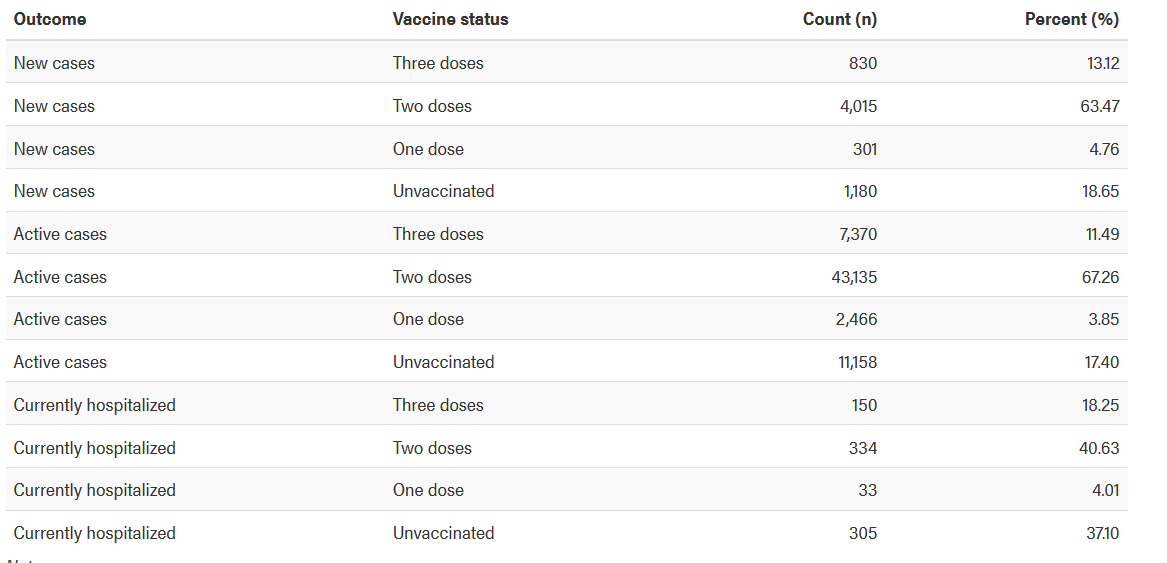
Source: https://www.alberta.ca/stats/covid-19-alberta-statistics.htm#vaccine-outcomes
Concluding Remarks
A sad axiom is that those who fail to read and follow lessons learned from history are doomed to repeat the same mistakes.
Our use of vaccines in history has been one of humankind’s great achievements. Their development and use, however, comes with many lessons, the most important being that we need to ensure that all vaccines are safe and effective before employing them. Failing to follow the testing and approval process across all sectors of society before use has led to unplanned, unexpected, and at times deadly outcomes. It appears that we have NOT learned these lessons.
Emergency management of a pandemic never relies on the arrival of vaccines to end it. A fully tested vaccine, which takes 3-5 years to develop under emergency protocols, will arrive too late to remedy an evolving virus and resolve a pandemic.
We need to return to a wiser approach that has worked well before. Emergency management principles, doctrine and process provide a full, effective and robust response – reducing fear, protecting those most at risk, minimizing the impact of the virus on all others, and the continuance of life as normal as possible in all ways possible.
Too bad we have failed to learn our lessons from previous pandemics. The impacts of the failed “Canadian Deadly Response to COVID-19” will last at least 60 years.